A portable test to help people suspected of having concussions or mild traumatic brain injuries has received a boost from the FDA.
BRAINBox Solutions, a molecular diagnostics innovator dedicated solely to the field of brain injury that works extensively with the Fralin Biomedical Research Institute at VTC, announced it has been granted U.S. Food and Drug Administration Breakthrough Device Designation to speed development of a multi-modality product, which includes a blood-based test to aid in prognosis and diagnosis of mild traumatic brain injury (mTBI).
Virginia Tech, with the Fralin Biomedical Research Institute and its clinical partner Carilion Clinic, will serve as one of the national anchor research and clinical sites to validate the test, which combines injury-related blood-protein biomarkers with computerized neurological assessments at the point of care.
“The FDA granted breakthrough designation of this new diagnostic approach from BRAINBox Solutions because it recognized how it benefits patients with an injury, especially those at greatest risk, including young people, athletes, soldiers, and the elderly, to be able to accurately and rapidly diagnosis mild traumatic brain injury,” said Michael Friedlander, executive director of the Fralin Biomedical Research Institute and Virginia Tech’s vice president for health sciences and technology. “This effort represents a collaboration of creative scientists and physicians who are committed to the power of large-scale, rigorous scientific testing to advance the diagnosis of mild traumatic brain injury.”
The data obtained will determine whether the new diagnostic approach may become a standard of care to aid in the diagnosis and predict the likelihood and length of post injury symptoms for mild TBI.
“With more than 5 million patients in the U.S. each year evaluated for mild TBI in hospital emergency departments, there is an enormous need for better tools to objectively diagnose and manage this condition,” said Donna Edmonds, president and chief executive officer of BRAINBox Solutions. “The breakthrough designation underscores the potential of BRAINBox TBI to change clinical practice in concussion management and will accelerate our development program.”
Stephen LaConte, an associate professor at the Fralin Biomedical Research Institute, will lead efforts in Roanoke in collaboration with his clinical colleague, Damon Kuehl, an emergency medicine physician with Carilion Clinic’s emergency department, to determine whether the BRAINBox TBI test is effective.
“The idea that a traumatic brain injury could be mild is misleading because these injuries have multiple downstream effects on a person’s behavior and day-to-day functioning, especially if the diagnosis isn’t made,” said LaConte, who is also an associate professor in the Department of Biomedical Engineering and Mechanics in the Virginia Tech College of Engineering. “The problem is mTBI is difficult to detect, even though it accounts for most brain injuries.”
The goal of the FDA’s Breakthrough Devices Program is to provide patients and health care providers with timely access to new medical devices by speeding up their development, assessment, and review, according to the FDA. The program offers manufacturers an opportunity to interact with FDA experts to efficiently address topics as they arise during the premarket review phase.
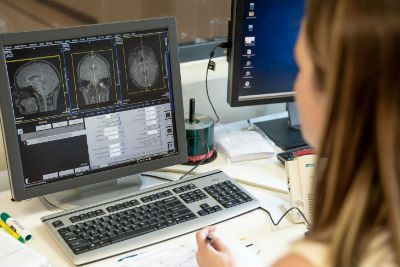
Currently, the state-of-the-art method for diagnosing mild traumatic brain injury is limited to neurocognitive tests. Now, by evaluating cognitive performance while a person is undergoing advanced brain imaging, researchers will determine the accuracy and relationship of blood biomarkers indicating brain injury very soon after the injury and at times afterward.
“A molecular test to identify traumatic brain injury in patients would be a breakthrough for care in an emergency setting,” said Damon Kuehl, who is also an associate professor at the Virginia Tech Carilion School of Medicine. “The current tools for diagnosing TBI are limited right now. A quicker, more definitive diagnosis would help us get patients the care they need faster.”
The BRAINBox TBI test will provide health care providers with a single score that incorporates the data from all the test components and with additional data useful for guiding treatment.
“As part of a specific site sub-study, Dr. LaConte will deploy sophisticated structural and functional brain imaging technologies in conjunction with clinical assessments by Dr. Kuehl to evaluate BRAINbox’s blood-based biomarker assay,” Friedlander said. “When integrated with a battery of neuropsychological tests and through standardization across multiple sites, this approach offers an exciting new opportunity for the development of a true point of care objective assessment for diagnosing mild traumatic brain injury. Such a potentially precise and objective diagnostic would also be a boon to the application of appropriate personalized therapeutics for individual patients.”
Faculty with the University of Virginia Health System will perform parallel diagnostic blood tests, neuroimaging, and neuropsychological studies as part of the collaborative program to evaluate BRAINbox’s multi-modality product.
In addition, Virginia Tech’s athletic departments, with Virginia Tech football team physician Gunnar Brolinson, the vice provost for research at the Edward Via College of Osteopathic Medicine, are expected to recruit volunteers for the research program.
In 2018, the project won the support of the Virginia Catalyst, formerly known as the Virginia Biosciences Health Research Corp., which has awarded a $500,000 grant to the Fralin Biomedical Research Institute at VTC to further develop and commercialize the multi-modality system.